Citation: Saito-Morooka F (2014) The prevalence of the parasitic nematode Sphaerularia sp. in the overwintering gynes of Parapolybia spp. (Hymenoptera, Polistinae). Journal of Hymenoptera Research 38: 37–43. doi: 10.3897/JHR.38.6562
I report for the first time the parasitization of overwintering gynes of Parapolybia species by the entomogenous nematode Sphaerularia sp. (Tylenchidae). The nematode was found in 42% of the overwintering wasp clusters examined but occurred in only 6% of individual wasps. The prevalence of parasitic nematodes among group hibernating hymenoptera is briefly discussed and compared with that in solitary hibernating hymenopterans.
Parapolybia, group hibernation, gyne, parasitic nematode, Polistinae
The parasitic nematodes of social hymenopterans include Mermithidae, Pheromermis pachysoma (von Linstow), for Vespula spp. (
During their overwintering diapause, gynes of bees and hornets are prone to nematode infection by the adult infective form (= inseminated females). By the following spring, the uterine sacs of parasitic nematode females are developed. Instead of founding a colony, parasitized overwintered gynes look for potential hibernation sites and parasitic juveniles are released from the host’s metasoma into hibernacula by late summer (
In temperate regions, social hymenopterans have two principal modes of hibernation (but Apis spp. do not hibernate): independently or in groups. In Bombus, Vespa, and some Polistes species a single gyne, or few gynes, tunnel under tree bark, soil, or rotten wood before winter and diapause until the following spring (
This is the first report of the infection of gynes of the group hibernating polistine wasp Parapolybia indica by parasitic nematodes. In this study, I describe the prevalence and condition of wasps infected by parasitic nematodes in their overwintering clusters and briefly compare them with solitary hibernating hymenopterans.
Parapolybia wasps were collected in Japan from October 2009 to April 2010 in the forest park of Mito city, Mito (36°25'N, 140°22'E, alt. 130 m, site A), Ibaraki Botanical Garden, Naka (36°29'N, 140°26'E, alt. 60 m, site B), Hitachi Ohmiya (36°39'N, 140°16'E, alt. 160 m, site C), the Seminar House of Ibaraki University, Daigo (36°49'N, 140°23'E, alt. 190 m, site D), Ibaraki Prefecture; in October 2012 in Rissho University, Kumagaya (36°06'N, 139°22'E, alt. 50 m, site E), and Nourin park, Fukaya (36°06'N, 139°17'E, alt. 90 m, site F), Saitama Prefecture. Wasps were collected in the following four phases: phase I) the pre-hibernating phase, which occurs shortly after mating and colony break up, in which females temporarily gather at a nest (possibly at their natal nest) or on plant leaves from late August to mid-October (sites D, E, and F); phase II) the early hibernating phase, in which females alternate between hibernacula from late October to mid-November (sites A and D); phase III) the late hibernating phase, in which females remain in the hibernaculum from mid-January to early February (sites A, B, and C); and phase IV) the precolony founding phase, which occurs shortly after emergence of gynes from hibernation in April (sites A and B).
Most overwintering wasps were found in or near hollows of broadleaf trees such as Quercus serrata Murray, Quercus acutissima Carruthers, or Prunus jamasakura Siebold, except for cluster no. A-1, which was occurred under wooden stairs in the park. Wasps resting around the entrances of the holes were collected with forceps during the phase II to IV at sites A and B. For wasps deep inside a tree hollow, the tree was cut off or the entrance of the hollow was bored out, and wasps were extracted with long forceps and/or a wire during phase II at site D and during phase III at sites A and B. The collected wasps were transported in a cool box with ice and stored in that box until dissection later the same day.
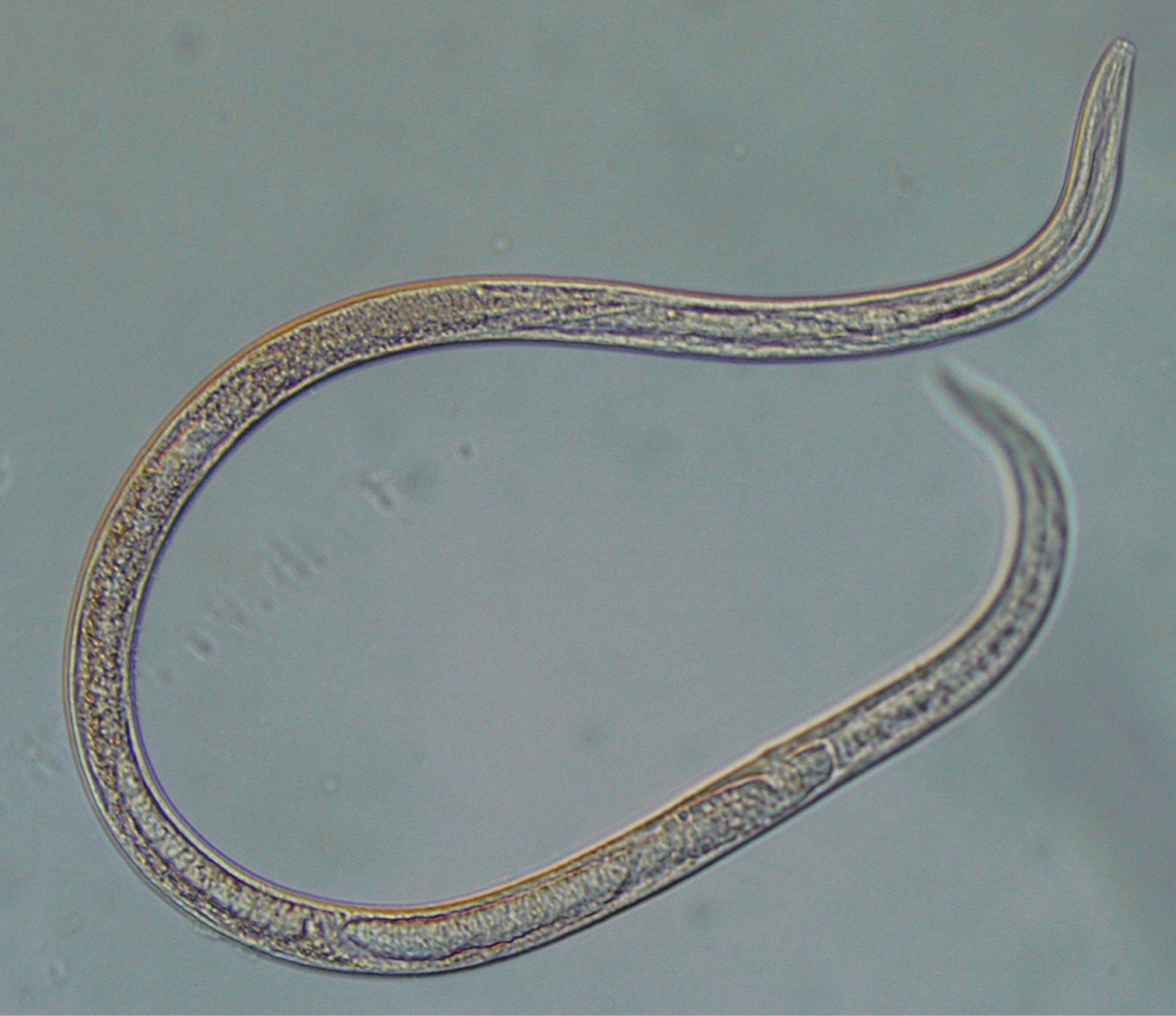
The metasomas were removed from live wasps, or wasps killed by 60% ethanol, and dissection was performed on a glass Petri dish under a binocular microscope. The metasomas were examined for the presence of nematodes. Parasitic nematodes were categorized into the following three stages: juveniles (small individuals with of indeterminate sex); uteria (mature adult females with developed uterine sacs); and infective forms (adult females without such sacs) (Fig. 1). Voucher specimens of nematodes and wasps were deposited in the Natural History Collection of Ibaraki University.
Photomicrograph of a parasitic nematode found in Parapolybia. It is considered a free-living or infective form of nematode because it lacks a well-developed everted uterus sac.
In addition, wasps were examined for the presence of fat bodies, sperm in the spermatheca, and the degree of wing wear. Wasps with fat bodies, sperm in the spermatheca, and a lack of wing wear were classified as gynes, whereas individuals lacking fat bodies or sperm in the spermatheca, and with worn wings were classified as workers.
For Parapolybia indica, three pre-hibernating clusters (gathered at the natal nest) in groups of 14, 16 and 99 (129 total individuals) were collected. And an additional 551 wasps were collected in phases II-IV from 23 additional hibernacula in the phase II–IV (Table 1). Solitary hibernating wasps were found at sites A, B, C, and D (n = 5). The number of individuals found in a hibernating cluster varied from 2 to 192 (n = 18; mean ± SD = 30.44 ± 44.98, median = 11).
The number of collected overwintering clusters and wasps. The number of clusters/wasps infected with Sphaerularia sp. is shown in parentheses. The breakdowns of the number of infected wasp individuals in each overwintering phase are shown in four columns on the right side.
| Species | Site | Location | No. of clusters | No. of wasps | Overwintering phase | |||
|---|---|---|---|---|---|---|---|---|
| Phase I | Phase II | Phase III | Phase IV | |||||
| Parapolybia indica | A | 36°25'N, 140°22'E, alt. 130 m | 17 (8) | 408 (28) | - | 231 (14) | 138 (11) | 40 (3) |
| B | 36°29'N, 140°26'E, alt. 60 m | 2 (1) | 63 (7) | - | - | 55 (5) | 8 (2) | |
| C | 36°39'N, 140°16'E, alt. 160 m | 1 (0) | 1 (0) | - | - | 1 (0) | - | |
| D | 36°49'N, 140°23'E, alt. 190 m | 4 (2) | 93 (7) | 14 (0) | 79 (7) | - | - | |
| E | 36°06'N, 139°22'E, alt. 50 m | 1 (0) | 99 (0) | 99 (0) | - | - | - | |
| F | 36°06'N, 139°17'E, alt. 90 m | 1 (0) | 16 (0) | 16 (0) | - | - | - | |
| total | 26 (11) | 680 (42) | 129 (0) | 310 (21) | 193 (16) | 48 (5) | ||
| Percentage | 42.3% | 6.2% | 0.0% | 6.8% | 8.2% | 10.4% | ||
| Parapolybia varia | D | 36°49'N, 140°23'E, alt. 190 m | 1 (0) | 102 (0) | ||||
After dissection, thirteen individuals of Parapolybia indica were classified as workers (site A, n = 4; site D, n = 1; site E, n = 7; site F, n = 1). They were collected in phases I-II and had no parasitic nematodes in their metasomas. Seven wasps collected before the phase III had no fat bodies or sperm in the spermatheca, and their wings were slightly damaged (site A, n = 5; site D, n = 2). For such samples, the wings may have been damaged during collection, and/or they were recently emerged workers. The other Parapolybia indica (n = 660) and Parapolybia varia (n = 102) had fat bodies, sperm in the spermatheca, and a lack of wing wear and were classified as gynes.
The total number of individuals infected by parasitic nematodes was 42. Such individuals were collected from their hibernacula during phases other than the phase I (Table 1). No phase I wasps were found to be nematode infected. All infected individuals were Parapolybia indica and were clearly classified as gynes. The prevalence (%) of parasitic nematodes in each overwintering cluster varied from 3.5 to 50.0 (median = 10.5). No statistical relationship was found between the cluster size and prevalence (correlation efficient r = -0.14, R ver.2.15.2,
A single overwintering cluster of Parapolybia varia was found at site D. It was without nematode infection. This cluster was found in the same tunnel as a cluster of Parapolybia indica. However, each cluster was monospecific and no species mixing was observed in the tunnel. Thus, in this study, only individuals of Parapolybia indica were infected by nematodes.
Parasitic nematodes were found only in the hemocoel. The stages of nematodes found in the wasp metasomas varied based on the phases of the wasps at collection. Infected wasps collected from phase II had a large number of juveniles and one or two uteria in their abdomen. Uteria and infective forms (Fig. 1) were found in the metasomas of wasps from phase III clusters. Infected wasps from phase IV contained only uteria.
For Vespa simillima, more than 60% of overwintered gynes were parasitized by Sphaerularia vespae (
Infection of hosts (bees and hornets) by parasitic nematodes of the genus Sphaerularia occur during the overwintering season in hibernacula such as soil and rotten wood (
The present study was supported by grants from the Japan Society for the Promotion of Science (no. 08J03623). I thank N. Kanzaki for the identification of parasitic nematodes.